
The active ingredient in Infuse Bone Graft-rhBMP-2-is a manufactured version of a protein already present in the body that promotes new bone growth. For these surgeries, it can offer several benefits, including eliminating the need to harvest bone from the patient's body in a secondary procedure, shorter operating times, and proven rates of fusion or bone formation. Infuse Bone Graft is FDA-approved for certain spine, oral-maxillofacial and orthopedic trauma surgeries. "We believe in the safety and effectiveness of Infuse Bone Graft and reiterate our continued commitment to the product." "With these expanded indications, we can bring the benefits of this important technology to more patients to help ensure they achieve a solid fusion and the best potential for a positive clinical outcome," said Doug King, senior vice president and president of Medtronic's Spinal business, which is part of the Restorative Therapies Group at Medtronic. This condition can cause back and/or leg pain, as well as functional problems, such as tingling or numbness in the legs or buttocks or difficulty walking. Infuse Bone Graft is used with certain Medtronic interbody fusion devices to treat lumbar degenerative disc disease. 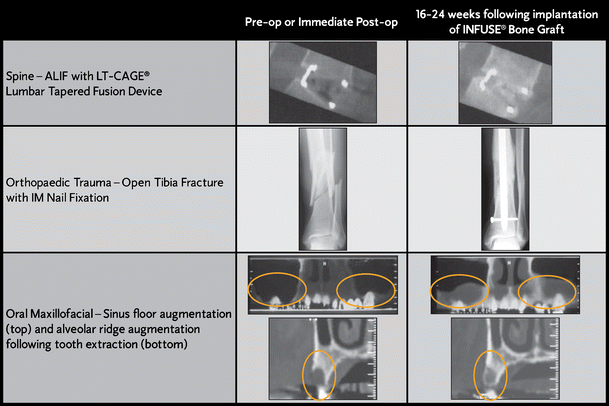
"In particular, I believe that the proven osteoinductive properties of Infuse Bone Graft are particularly beneficial when used in conjunction with PEEK interbody spacers and the less invasive OLIF technique, which circumvents the psoas muscle and minimizes disruption of the surrounding soft tissues and neural structures." "For my anterior and anterolateral lumbar spine fusion cases, the use of Infuse Bone Graft allows me to reliably obtain a solid arthrodesis without having to harvest bone from the patient's own hip which generally requires a second incision, results in significant pain, and increases the risk of complications such as bleeding or infection," said Peter Whang, MD, FACS, an associate professor in the Department of Orthopaedics at the Yale School of Medicine in New Haven, CT. Use in ALIF procedures with certain sizes of the PEEK Perimeter Implant at a single level from L2-S1. Use in OLIF25 Procedures with certain sizes of the PEEK Clydesdale Implant at a single level from L2-L5. Use in OLIF51 Procedures with certain sizes of the PEEK Perimeter Implant at a single level from L5-S1. The new indications for Infuse Bone Graft are: With this expanded approval, Medtronic will be able to market Infuse Bone Graft for use with certain spine implants made of polyetheretherketone (PEEK) in oblique lateral interbody fusion (OLIF) and anterior lumbar interbody fusion (ALIF) procedures. Upon receiving final labeling approval from the FDA, the company expects to begin marketing these expanded indications in early calendar year 2016. Food and Drug Administration (FDA) approval of additional spine surgery indications for Infuse Bone Graft. In total, authors of the published medical studies were also found to have been paid $210 million by Medtronic for unrelated work between 19.Medtronic plc has announced U.S. The Senate report found that Medtronic had been deeply involved in the drafting, editing and shaping the content that went into the medical journal articles that had encouraged off-label use of the bone grafts. In an effort to learn more about Medtronic’s promotion of Infuse Bone Grafts for off-label purposes, the Senate Finance Committee produced a report based on thousands of documents from Medtronic to investigate a numberof published medical studies that encouraged doctors to begin using the bone grafts for off-label surgeries. If an injury was suffered as a result of an Infuse Bone Graft, on or off-lable, you may have grounds for an Infuse Bone Graft lawsuit. Such unapproved or off-label uses have caused serious and sometimes life-threatening side effects. Although the FDA approved the Infuse Bone Graft for use in lower spine-repair surgery to promote bone growth, Medtronic allegedly promotes its use to doctors for non-FDA-approved surgeries including cervical fusion or lumbar fusion. Life-threatening injuries are caused by Medtronic’s Infuse Bone Graft, especially after doctor’s use it in off-label surgeries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
